States of Matter - Online Test
Q1. Calculate the volume occupied by 8.8 g of CO2 at 31.1 and 1 bar pressure. R = 0.083 bar .
Answer : Option B
Explaination / Solution:
V=wRTMPV=8.8×0.083×304.144×1=5.05L
Q2. 2.9 g of a gas at 95 occupied the same volume as 0.184 g of dihydrogen at 17 , at the same pressure. What is the molar mass of the gas?
Answer : Option A
Explaination / Solution:
PV=(/M)R where m 1 =2.9g and =368K PV= (/2)where =0.184 and = 290K
Q3. A mixture of dihydrogen and dioxygen at one bar pressure contains 20% by weight of dihydrogen. Calculate the partial pressure of dihydrogen.
Answer : Option C
Explaination / Solution:
P=P1+P2 (Daltons Law of partial pressure).
Q4. The vapor pressure of different substances at the same temperature increases if
Answer : Option A
Explaination / Solution:
if the intermolecular forces of the substance are weak then vapour pressure increases.
Q5. A person living in Shimla observed that cooking food without using pressure cooker takes more time. The reason for this observation is that at high altitude:
Answer : Option C
Explaination / Solution:
As we move to higher altitude P decreases.
Q6. Which of the following property of water can be used to explain the spherical shape of rain droplets?
Answer : Option C
Explaination / Solution:
surface Tension is used.
Q7. A plot of volume (V ) versus temperature (T ) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in Figure. Which of the following order of pressure is correct for this gas?
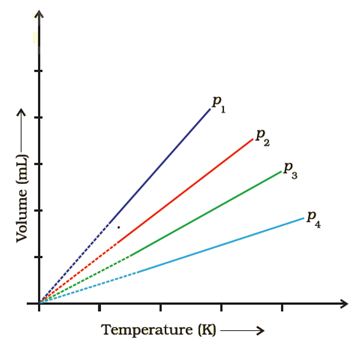

Answer : Option B
Explaination / Solution:
keep Temperature constant we know Pα1/V So as V increases P decreases.
Q8. The interaction energy of London force is inversely proportional to sixth power of the distance between two interacting particles but their magnitude depends upon
Answer : Option A
Explaination / Solution:
Its magnitude depends on the polarisability of interacting particles.
Q9. Dipole-dipole forces act between the molecules possessing permanent dipole. Ends of dipoles possess ‘partial charges’. The partial charge is
Answer : Option A
Explaination / Solution:
It is less than unit electronic charge.
Q10. As the temperature increases, average kinetic energy of molecules increases. What would be the effect of increase of temperature on pressure provided the volume is constant?
Answer : Option C
Explaination / Solution:
P α T at constant volume.This is according to Gay Lussac’s law-“ At constant volume, the pressure of a given mass of a gas is directly proportional to its absolute temperature.”
