Organic Chemistry Some Basic Principles and Techniques - Online Test
Q1. Covalent bonds are formed with other carbon atoms due to
Answer : Option D
Explaination / Solution:
Catenation is the linkage of atoms of the same element into longer chains.Catenation occurs most readily in carbon, which forms covalent bonds with other carbon atoms to form longer chains and structures. This is the reason for the presence of the vast number of organic compounds in nature.
Q2. Which of the following organic compound Wohler synthesised from an inorganic compound, ammonium cyanate in the presence of heat:
Answer : Option B
Explaination / Solution:
The reaction can be given as starting with solutions of potassium cyanate and ammonium chloride which are mixed, heated and cooled again to get ammonium cyanate salt which on decomposition gives the urea.
Q3.
Straight or branched chain compounds, for example, the below ones are called
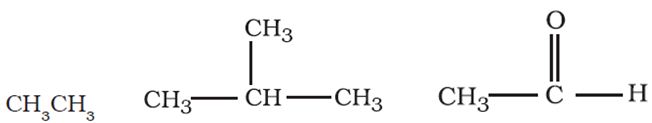
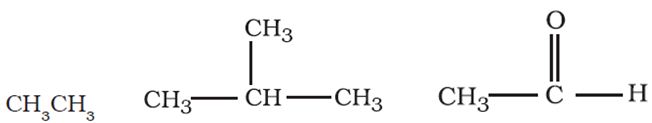
Answer : Option C
Explaination / Solution:
They are straight chain compounds known as aliphatic comounds.
Q4. Correct IUPAC name the following compound is
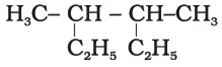
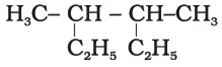
Answer : Option D
Explaination / Solution:
3,4- dimethylhexane will be correct iupac name as 6 membered carbon chain will be the main chain.
Q5. Benzene and other related ring compounds (benzenoid) such as given below are called:
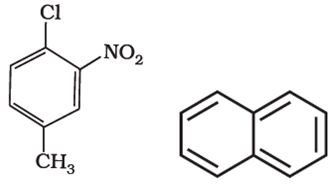

Answer : Option C
Explaination / Solution:
they are aromatic system as they have benzene ring intact.
Q6. The following compound is:
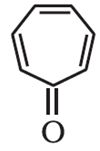

Answer : Option D
Explaination / Solution:
Tropone or 2,4,6-cycloheptatrien-1-one is a non-benzenoid aromatic compound. The compound consists of a ring of seven carbon atoms with three conjugated alkene groups and a ketone group.
Q7. Compounds containing carbon atoms joined together in the form of a ring as following are:
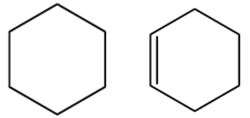

Answer : Option C
Explaination / Solution:
An alicyclic compound is an organic compound that is both aliphatic and cyclic. They contain one or more all-carbon rings which may be either saturated or unsaturated, but do not have aromatic character.
Q8.
The molecular formula represents two alcohols: propan-1-ol and propan-2-ol. This property is called as:
Answer : Option D
Explaination / Solution:
propan-1-ol and propan-2-ol are positional isomers as they differ only in the position of functional group -OH.
Positional isomers are isomers that have the same carbon skeleton and the same functional groups but differ from each other in the location of the functional groups on or in the carbon chain.
Q9. What is the correct order of decreasing stability of the following cations.
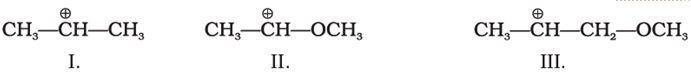
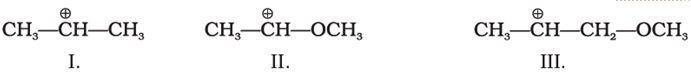
Answer : Option B
Explaination / Solution:
II is very stable because +ve charge is dispersed by +R effect of
Q10. What is the type of hybridisation of each carbon in the compound
Answer : Option B
Explaination / Solution:
C in CH3 is and C of C=O bond is .
